4. Long term impacts: How infection and immunity affect tissue function persistently
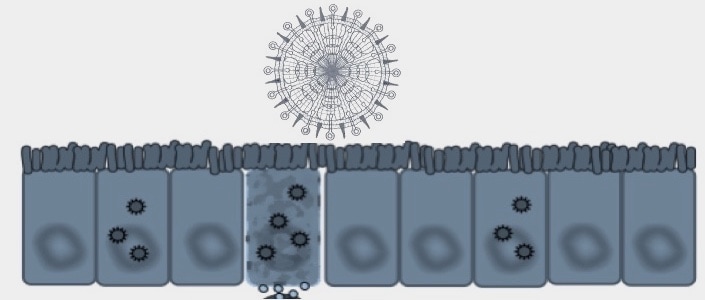
Both infections and the immune responses they trigger cause collateral damage to the infected tissue. This is especially important in organs like the lung. The structure of the lung is exquisitely designed to facilitate gas exchange. Lung infection causes dramatic changes in lung morphology, including an immune cell influx, thickening of alveolar walls and initiation of new lymphoid tissues. These changes are accompanied by an extensive programme of remodelling and repair, essential to restore normal lung structure and maintain breathing. We are interested in the instructions and decisions that direct these morphological changes, and how they can be influenced by both the infection and the consequent immune response.
|
One of our goals is to understand the cytokine signals that optimise the host:pathogen interaction in influenza infection. In this project, our objective is to establish how the immune system interacts with stromal cells in the lung to promote effective remodelling and repair. We are currently focused on basal cells, key progenitor cells for the alveolar epithelium, and cells that are surprisingly both infected by influenza virus and likely to survive the infection. We want to know whether the virus can alter the basal cell, changing the differentiation patter of daughter cells; and/or whether inflammatory or anti-inflammatory cytokines released during the immune response to infection can achieve a similar effect.
|
Such changes are likely to be long-term changes, permanently altering the phenotype of the regenerated lung epithelium. Such changes have been called "innate memory" or "trained immunity", examples of memory in the stromal or innate immune compartments. We are also investigating other examples of trained immunity in the lung, after influenza virus infection, including changes in macrophage dynamics. These macrophage studies are a collaboration with Dr Steve Jenkins and his team at the University of Edinburgh. Together our data are providing new insight into the mechanisms that drive pathology and recovery during and after an acute influenza infection.